Abstract
Background: In November 2020, the U.S. Food and Drug Administration (FDA) issued emergency use authorization (EUA) for monoclonal antibody (mAb) therapy in patients with mild to moderate COVID-19 who are at high risk for disease progression. These mAbs reduce the risk of hospitalization in the general population. However, its efficacy and safety in immunocompromised hematology patients are not known.
Methods: From November 9th, 2020, until February 28th, 2021, all adult hematology patients with mild to moderate COVID-19 disease who received monoclonal antibodies within 10 days of symptoms onset were included. Patients who were asymptomatic, had severe or critical COVID-19 disease, or were hospitalized at the time of COVID-19 diagnosis were excluded. Baseline demographic, clinical outcomes, and hematologic-related data were extracted. All statistical analysis was performed using SAS statistical software.
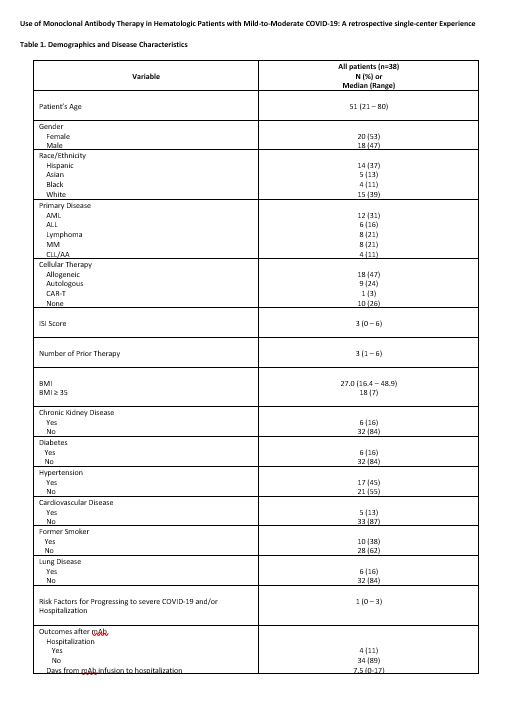
Results: Thirty-eight hematology patients with mild to moderate COVID-19 disease who received mAb therapy under EUA were included in this study. Thirty (79%) patients received bamlanivimab and 8 (21%) casirivimab-imdevimab. Baseline characteristics prior to mAB administration include: 53% female, median age of 51 years (range: 21-80), with 18% above 65 years old. Twenty-eight (74%) patients received cellular therapy: 18 (47%) had undergone allogeneic hematopoietic cell transplantation (HCT), 9 (24%) autologous HCT, and 1 (3%) chimeric antigen receptor T-cell (CAR T) therapy. Among the 17 patients who had COVID-19 disease after HCT, the median time to COVID-19 diagnosis was 22.8 months (range: 2.6-274.4) from HCT to COVID-19 diagnosis. Twelve out of 17 (71%) alloHCT patients were being managed for active graft-vs-host disease (GvHD) at the time of COVID-19 diagnosis (chronic GVHD: n=11 [mild: 4, moderate: 4, severe: 3], acute GVHD (grade 2): n=1). Ten (59%) alloHCT patients were on immunosuppressant therapy at the time of COVID-19 diagnosis. Fifteen (39%) patients were on active treatment for their hematologic malignancy (HM) at the time of COVID-19 diagnosis with a mean of 3 previous lines of treatment (range: 1-6). Additional patient characteristics are shown in Table 1.
mAb therapy under EUA was well tolerated in this patient population with only 1 (3%) patient having experienced an adverse reaction characterized as headache.
Four (11%) patients were hospitalized due to COVID-19, and 2 (5%) progressed to severe disease. All four patients had received bamlanivimab. The median time for hospitalization from diagnosis of COVID-19 to admission date was 8 days (range: 1-20) while median time from mAB infusion to hospitalization was 7.5 days (range: 0-17). One patient (3%) died within 30 days of COVID-19 diagnosis; the cause of death was COVID-19 disease.
Most patients (n=34, 89%) ultimately tested negative for SARS-CoV-2 by PCR after mAb infusion. 34% of patients (n=13) cleared the virus within 2 weeks of receiving mAb infsuion. The median time to clearance of viral shedding was 25.5 days (range: 7-138). After mAb infusion, most patients (10/15; 67%) who were previously on active treatment for HM prior to diagnosis of COVID-19 resumed therapy for their HM with a median delay of 21.5 days (range: 12-42).
We observed a significant difference in hospitalization was amongst patients who received a HCT vs. non-HCT (0%, 0/26 and 36%, 4/11 respectively; p<0.01). None of the other patient characteristics, which included: gender, ethnicity, age, BMI, smoking, obesity, chronic kidney disease, diabetes mellitus, hypertension, coronary vascular disease, and lung disease, were associated with significantly increased rate of hospitalization.
Conclusion: This study demonstrates that SARS-COV2 specific mAb use in malignant hematology patients under EUA was safe and may reduce hospitalization as reported in the literature amongst those at high risk for disease progression. Thus, the access to SARS-COV2 mAb in this population who is at increased risk for complications from SARS-COV2 infection is critical in reducing progression to severe COVID-19 disease and hospitalization.
Ali: Incyte: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; CTI BioPharma: Membership on an entity's Board of Directors or advisory committees; BMS: Speakers Bureau. Aribi: Seagen: Consultancy. Artz: Radiology Partners: Other: Spouse has equity interest in Radiology Partners, a private radiology physician practice. Koller: Novartis: Consultancy. Nikolaenko: Rafael Pharmaceuticals: Research Funding; Pfizer: Research Funding. Shouse: Beigene: Honoraria; Kite Pharma: Speakers Bureau. Stein: Amgen: Consultancy, Speakers Bureau; Celgene: Speakers Bureau; Stemline: Speakers Bureau. Marcucci: Abbvie: Other: Speaker and advisory scientific board meetings; Novartis: Other: Speaker and advisory scientific board meetings; Agios: Other: Speaker and advisory scientific board meetings. Forman: Mustang Bio: Consultancy, Current holder of individual stocks in a privately-held company; Lixte Biotechnology: Consultancy, Current holder of individual stocks in a privately-held company; Allogene: Consultancy. Dadwal: AlloVir: Research Funding; Merck: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Astellas: Speakers Bureau; Shire/Takeda: Research Funding; Aseptiscope: Consultancy; Janssen: Other: Investigator; Karius: Other: Investigator. Al Malki: CareDx: Consultancy; Rigel Pharma: Consultancy; Jazz Pharmaceuticals, Inc.: Consultancy; Neximmune: Consultancy; Hansa Biopharma: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal